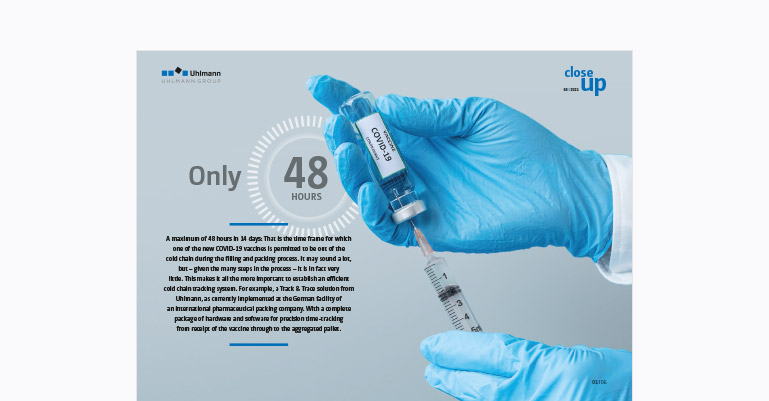
Wherever you look in the media today, the topic is always the same: The delay in getting the COVID-19 vaccines ready and the question: What is taking so long? The question should in fact be: How has everybody involved managed to get such a complex end-to-end process up and running in such a short space of time? Georg Schick, Global Product Manager of Track & Trace, answers this question on behalf of Uhlmann: “With a combination of experience, excellent coordination, the right software and hardware tools and, not least, the odd night shift.”
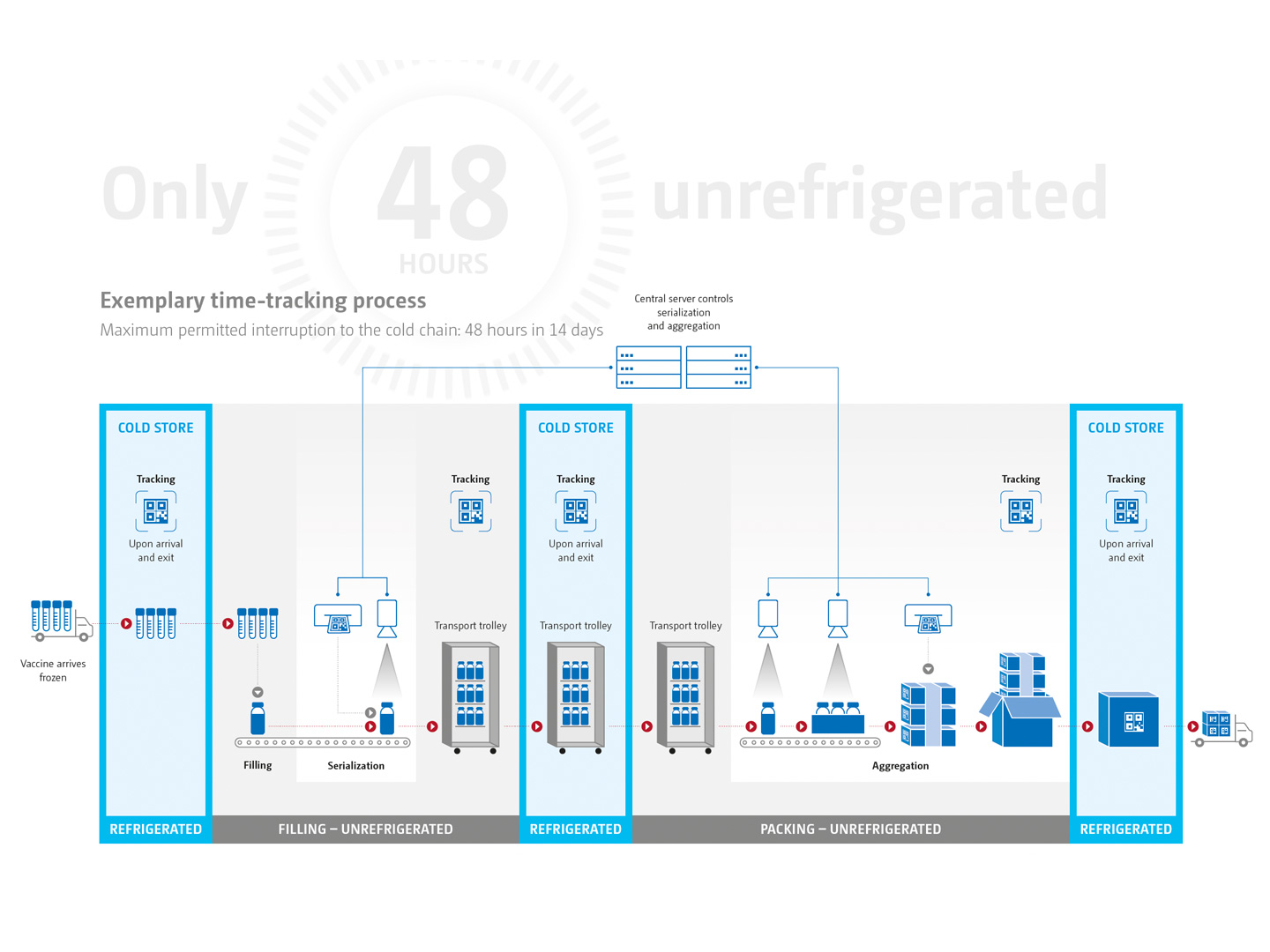
Medicine products in particular are very susceptible to outside influences. Moisture, cold or heat can compromise a product‘s effectiveness, as can excessive interruptions to the cold chain. To reliably identify these interruptions and to record the exact length of them, the Track & Trace specialists at Uhlmann have developed a complete solution. This allows for timings to be tracked throughout the entire process – from the delivery of the vaccine to the final freezing of the vials packed inside folding cartons and transit cases. And at the same time, it facilitates an aggregation from product to pallet following the principle of the parent-child relationship.
See the time at a glance, at any time

The key to success? Without doubt, it‘s our internally developed, modular software. This enables us to design the perfect configuration for the task – from the machinery to line management and even site management.
Steffen Beer, Project Manager Digital Solutions
Under intense pressure.
In August 2020 the Track & Trace specialists at Uhlmann received an inquiry; by the end of October the contract was in place. Since then, five employees from programming, electrical design and mechanical design have been working tirelessly on creating the solution for tracking the COVID-19 vaccine. What is special about this? While most Track & Trace solutions are integrated in Uhlmann packaging lines, the emphasis here is clearly on the software. According to Steffen Beer, Project Manager Digital Solutions, “software is always at the heart of a Track & Trace application. In this instance, however, our solution spans all the way from the receipt of goods to the filling process and right through to the packaging process.”
Continuous flow of data.
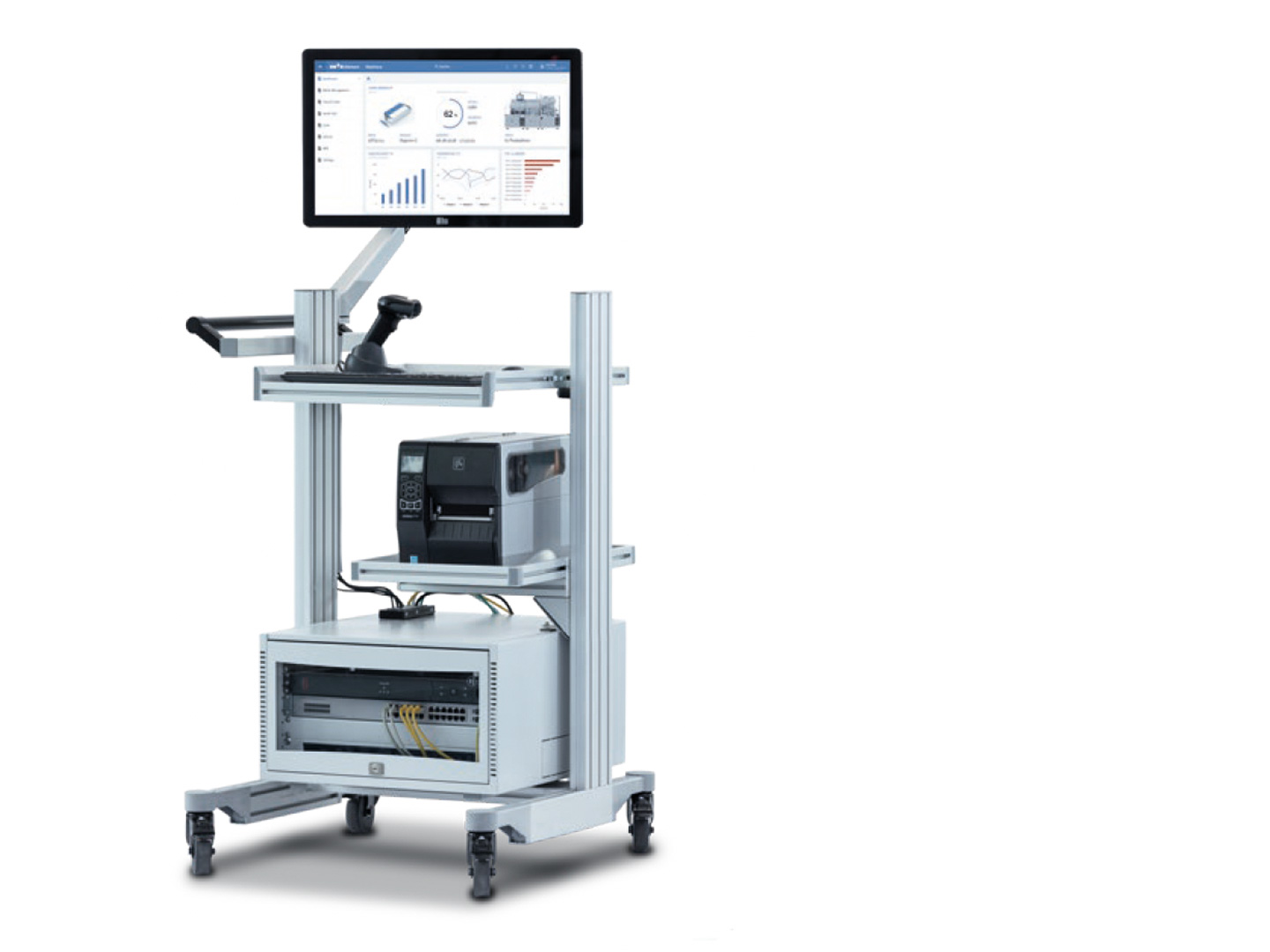
A central element of the application is a data server. This is where the batches to be tracked are created and finalized and where the reports are produced. Several Uhlmann Control Terminals T1 are integrated across the entire filling and packaging line and beyond for communication with the server. The terminals allow all the necessary data to be read and input in a user-friendly way. Once the filling process is complete, the products are packed on a cartoner, with each folding carton given a unique code. Another T1 terminal is integrated downstream for aggregation to the pallet.
A quick fix?
Customers had initially envisaged a temporary solution, with many of the steps being performed manually. This has since been rethought, however. The application is being designed for long-term use from the outset: The seria lization of the vials and the packing into folding cartons is undertaken by a cartoner, while the handling ahead of the cartoning process is performed by pick-and-place robots.
Right from the very beginning.
When the vaccine is received, the arrival time of each batch is recorded and the production order is issued … now the clock starts ticking! The batch takes the fastest route to the cold store, check-in and check-out at the filling station are recorded in the system by means of a time stamp.
A complex process, a one-stop solution

Whether it‘s time tracking or enhanced cold chain tracking, our clients benefit from the experience we bring, accumulated over many successful Track & Trace projects.
Georg Schick, Global Product Manager Track & Trace
Filling process with serialization.
At the filling station, the procedure is repeated: First comes the time stamp, then the job starts. Once the vials are filled with the vaccine, they are printed with a UV code which can then be read by the VisioRead camera system. Control Terminal T1 prepares and records the serialization data and the aggregation.
Enough time left for packing?
This is indicated at each scan by the traffic light system at the various terminals. Additionally, the times at which the product is unrefrigerated can be read from the browser at any time, including outside production.
Mission accomplished!
Has the traffic light system triggered no alarms? Have the sensitive products spent any unscheduled waiting time in the safety
of the cold store? If so, then the time tracking is successfully complete when the transit cases are registered in the freezer.
Not quite …
Steffen Beer‘s team are currently preparing everything for the FAT in April in Laupheim and the commissioning in May. For which, it‘s hoped, everyone will be present at the customer‘s site in order to make a thorough inspection of the application ”in the real world“.

Cold chain: The three levels of cold.
A cold chain is defined as “… the continuous system of refrigeration from manufacture through packaging to transport.” It is the system that ensures our salad leaves are fresh and crunchy when they reach our kitchen table. If the chain is broken, the goods can spoil and have to be thrown away. In the case of food products, this normally leads only to inconvenience. When it comes to medicines, however, too much disruption to the cold chain can have fatal consequences, and in some cases entire batches have to be destroyed; it can cause supply bottlenecks and, consequently, a shortage of supply for patients. This makes it all the more important that the prescribed temperature ranges are strictly maintained and that any interruptions required for processing remain within the specified range of tolerance. A cold chain splits into three levels – the colder, the more complex the requirements:
-
Refrigerated chain with temperatures of between
2 and 8 °C (fridge cold) e.g. for flu vaccines and insulin -
Frozen chain operating at -20 °C, e.g. for chicken pox vaccine
-
Ultra-low or deep-freeze cold chain
operated at -70 °C e.g. for Ebola and COVID-19 vaccines
Good reasons for cold chain tracking with Uhlmann.
Whether tracking medicines and medical devices in general or cold chain tracking, the ultimate aim is to provide extensive counterfeit protection, product safety and process reliability. The objective of Track & Trace by Uhlmann is to make the route there as easy and secure as possible. This is achieved with an end-to-end solution comprising machines, components, soft - ware, advice and services. For cold chain tracking within the packaging process and surrounding fields, Uhlmann offers two options:
- Time tracking
The tracking focuses solely on the time the temperature-sensitive products spend outside the cold chain. -
Cold chain tracking
This tracking provides precise records of the tem pe rature at any given time. This guarantees that the prescribed and defined temperature ranges are maintained at all times. This uses, for example, RFID temperature loggers, which are read out at relevant stations in the process.
If you‘d like to find out more, do not hesitate to e-mail Georg Schick at: schick.g@uhlmann.de