Sustainable pharmaceutical packaging: Forward-looking solutions for the pharmaceutical industry

The pharma packaging of the future is green and smart. It combines sustainability with smart technology – and is becoming the new standard in the pharmaceutical industry. No matter what stage of the development process for a new packaging solution you are at: As a technology leader in sustainable pharmaceutical packaging, we help you develop the right sustainable solution for your packaging.
Why sustainable packaging is becoming essential
From an industry trend to the new standard
For a long time, sustainable pharmaceutical packaging was viewed as a voluntary initiative within the pharmaceutical industry. Today, it represents a strategic area of innovation. This transformation is being driven by legal requirements as well as by rising expectations from partners, investors and the public. Companies that fail to make their packaging solutions more sustainable risk falling behind the competition – or, in extreme cases, even losing market access.
As a result, more and more companies are systematically reviewing their packaging solutions: How much material – and, more importantly, what kind – is actually needed? Where can process steps be made more efficient? How can transport volumes and warehouse space be optimized? Sustainability is therefore becoming a driver of operational improvements.
Companies that strategically embed such measures do more than just meet regulatory requirements. They specifically use environmental criteria to further develop products, processes and business models. As a result, the entire packaging system – from development and material selection through production and logistics to disposal – is becoming a key factor in long-term competitiveness and sustainable business practices.
Uhlmann’s 5R approach for sustainable packaging
The pressure to make pharmaceutical packaging more sustainable is growing – and with it, the complexity of design, materials and processes. Based on our many years of experience, we know how to translate these requirements into viable solutions. Our 5R approach brings together the key levers for achieving this.
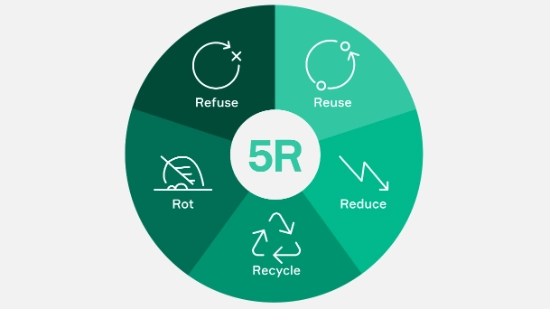
A look at the individual principles shows how this approach can be implemented in practice. Each of the 5Rs defines clear measures and helps to structurally embed sustainability throughout the entire product life cycle.
Sustainable packaging materials for pharma packaging
Play to your strengths – know your limits
Sustainable pharma packaging uses different types of materials, each with its own specific functional properties. These include recyclable plastics such as polypropylene (PP), high-density polyethylene (HDPE) and polyethylene terephthalate (PET), bio-based polymers, fiber-based materials and recycled plastics – i.e. plastics that have been mechanically or chemically recycled.
However, the choice of material is not primarily based on recyclability, but rather on the protection required for the medication. Barrier properties, stability and regulatory compliance set the framework. At the Packaging Competence Center, we evaluate these parameters in combination to determine the most suitable material for your specific packaging application.
„Sustainability will change pharmaceutical packaging and will pose new challenges for our customers. Specifically, it concerns recyclable packaging. The key is to balance sustainability with product safety, quality and GMP-compliant implementation.‟

Klaus Braig
Vice President Global Sales
Forward-looking solutions for the pharmaceutical industry
Sustainability consistently implemented
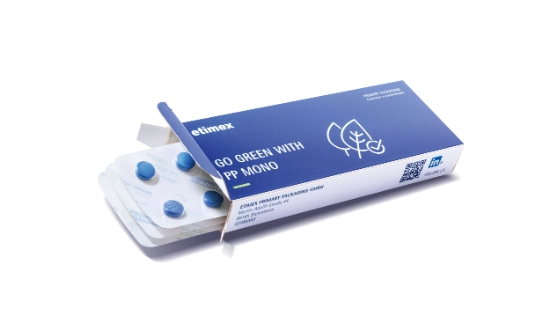
Mono-materials for pharmaceutical packaging
Multi-material composites – such as PVC/aluminum – have long been considered the standard in the packaging of solids as they offer impressive functional and economic advantages. However, their tightly bonded layers of material make recycling considerably more difficult. In light of this, the shift toward recyclable mono-material packaging is becoming a key focus of sustainable packaging strategies. Packaging made from a single material can be sorted by type and recycled.
We have been actively promoting the processing of mono-materials for many years and consistently tailor our machine technology to this end. For example, the BEC 400 is a compact, modular blister packaging line that reliably processes mono-PP films and produces corresponding blister packaging. We are also expanding our range of eco-friendly material options for the secondary packaging of parenteral products: Our BLU 200 is also packaged in mono-PP blisters.
Our packaging machines and lines can generally be designed or retrofitted to handle a variety of sustainable materials – including PET, HDPE and paper-based solutions, in addition to PP.
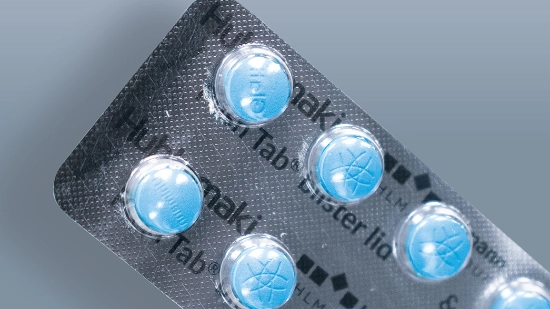
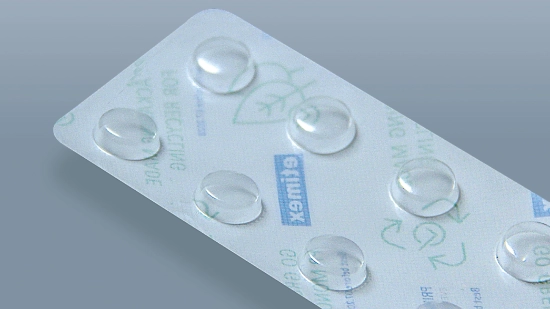
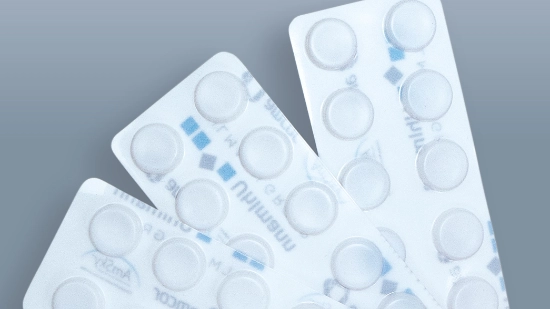
Cardboard trays for parenteral products
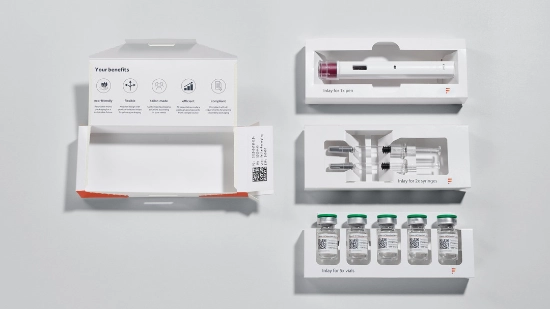
By using cardboard trays, we are systematically switching the secondary packaging for parenteral products to cardboard and replacing plastic while maintaining the same functionality. The slim, lightweight design reduces material usage and transport volume, is easy to recycle and remains sturdy at the same time. Delicate vials, syringes, ampoules and pens remain securely in place and protected. This reduces resource consumption, environmental impact and costs.
Our PTC 200 packaging line produces cardboard trays in various sizes and with different closure mechanisms – including medical device tracking. The packaging concepts take into account international sustainability and reporting frameworks such as CDP, UNGC, and GRI. The following illustrations show examples of different variations.
Our end-to-end service
From initial idea to finished product
Take advantage of our expertise to develop tailored sustainable packaging solutions and implement them efficiently and safely. At our Uhlmann Packaging Competence Center, we support you every step of the way – from the initial assessment to the seamless integration into your production process. The following six steps outline exactly how we go about this:
- Define requirements
In a workshop, we work with you to analyze your requirements and circumstances. - Research materials
We identify suitable packaging materials and concepts for your application. - Perform a life cycle assessment
We assess the environmental impact of potential packaging solutions. - Test the packaging
In our test center, we test materials, processes and machine parameters under realistic conditions. - Validate the solution
We validate the selected packaging solution and ensure process stability. - Oversee the start of production
We help you integrate the solution into your production process – whether by installing new machinery or modifying existing equipment.
Printing on sustainable materials
We are also here to assist you with any other needs you may have regarding sustainable packaging solutions. Our technologies also enable precise print mark and registered printing on sustainable packaging materials.